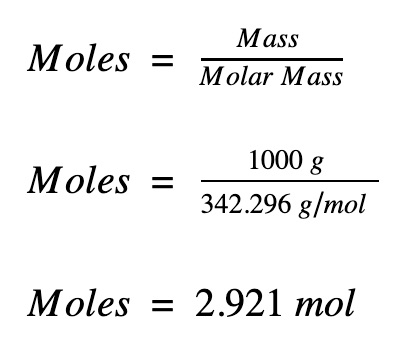How To Solve Most Mole Calculation Questions – Part 1 | O Level Chemistry Tuition | Leading Chemistry Tuition Centre

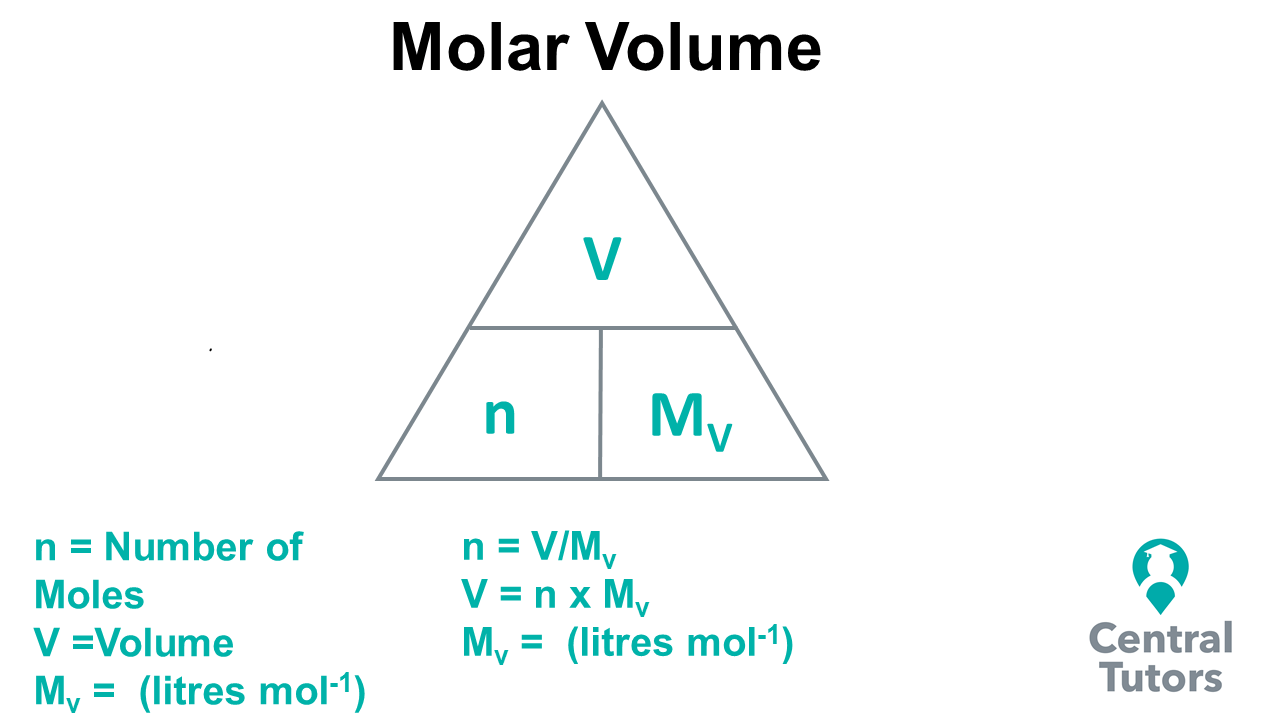
Calculations in Chemistry To calculate the number of moles in a solid we use the following Mole Triangle g n gfm g = Mass in Grams n= Number of moles gfm=gram. -

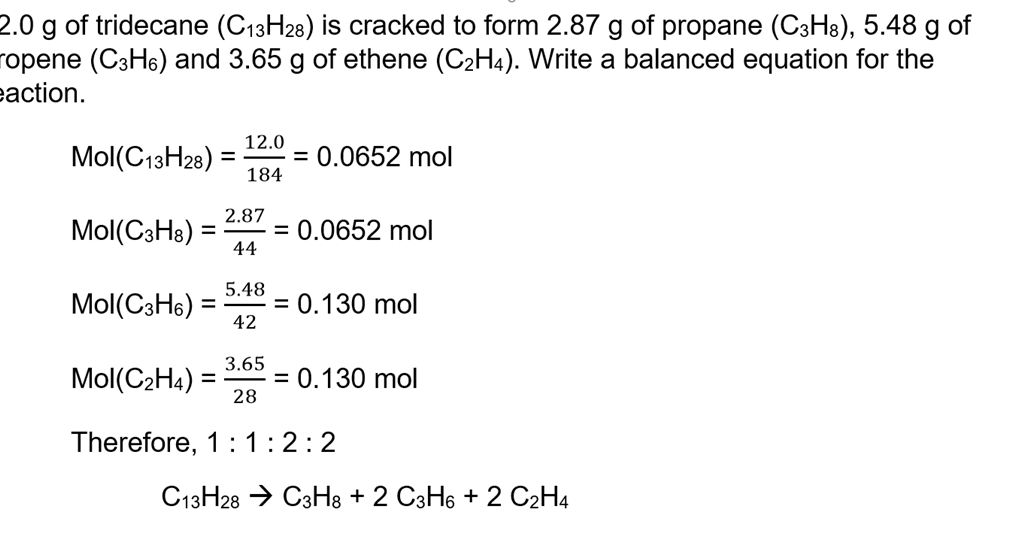
How to Find Moles of Product from Moles of Reactant using a Chemical Equation | Chemistry | Study.com